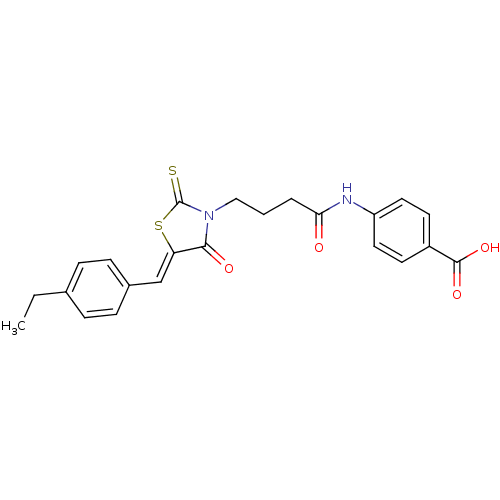
BDBM58651 4-[4-[(5Z)-5-(4-ethylbenzylidene)-4-keto-2-thioxo-thiazolidin-3-yl]butanoylamino]benzoic acid::4-[4-[(5Z)-5-[(4-ethylphenyl)methylidene]-4-oxidanylidene-2-sulfanylidene-1,3-thiazolidin-3-yl]butanoylamino]benzoic acid::4-[4-[(5Z)-5-[(4-ethylphenyl)methylidene]-4-oxo-2-sulfanylidene-1,3-thiazolidin-3-yl]butanoylamino]benzoic acid::4-[[4-[(5Z)-5-[(4-ethylphenyl)methylidene]-4-oxo-2-sulfanylidene-3-thiazolidinyl]-1-oxobutyl]amino]benzoic acid::MLS001075748::SMR000646238::cid_1985027
SMILES CCc1ccc(\C=C2/SC(=S)N(CCCC(=O)Nc3ccc(cc3)C(O)=O)C2=O)cc1
InChI Key InChIKey=PITVDYLNTHZLRC-RGEXLXHISA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 58651
Found 2 hits for monomerid = 58651
TargetTyrosine-protein phosphatase non-receptor type 22(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 7.68E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetTyrosine-protein phosphatase non-receptor type 22(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 7.60E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute(SBMRI, San Diego, CA...More data for this Ligand-Target Pair